-

Medical Device Management Ltd In collaboration with Technology Partnership
Quality and Regulatory Compliance
Software for the Medical Industry


In an ever-changing world, regulatory compliance is often seen as complex, costly and time-consuming.
Our philosophy is:
The MediqARC Quality Management System (QMS) has been developed to meet the requirements of organisations involved in the design, manufacture, service, maintenance or sterilisation of Medical Devices. From manufacturer to the hospital biomedical engineer and sterilisation department.
Based on ISO 13485 and 21 CFR part 820 MediqARC allows the documentation associated with the implementation, operation and maintenance of the QMS to be accessed, monitored and displayed from a single screen.
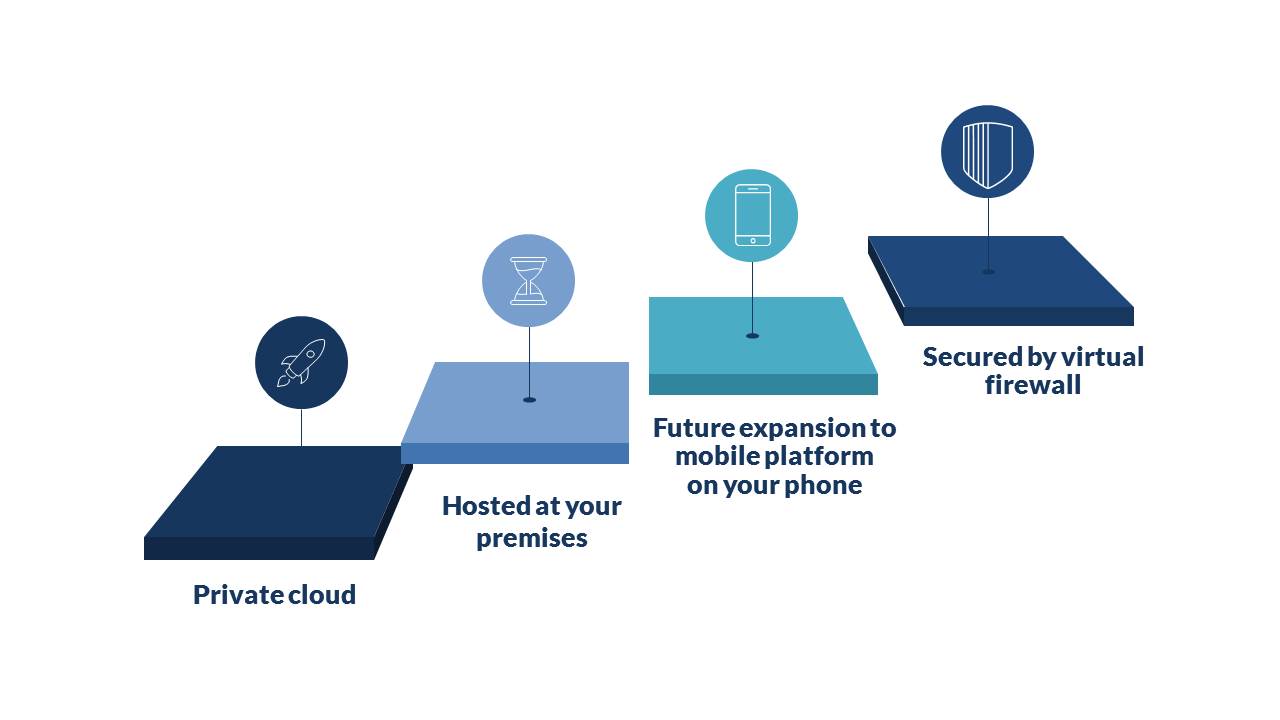
Private cloud provides global access ideal for working from multiple locations.
Hosted at your premises ensures access even when the internet is not available and increases privacy protection.
Inclusion of the mobile platform is ideal for those on the move and checking or entering information such as batch details or non-conformance while away from your desk in the warehouse or factory.
Secured by Virtual Firewall to reduce the risk of unauthorised access and to improved cyber security.
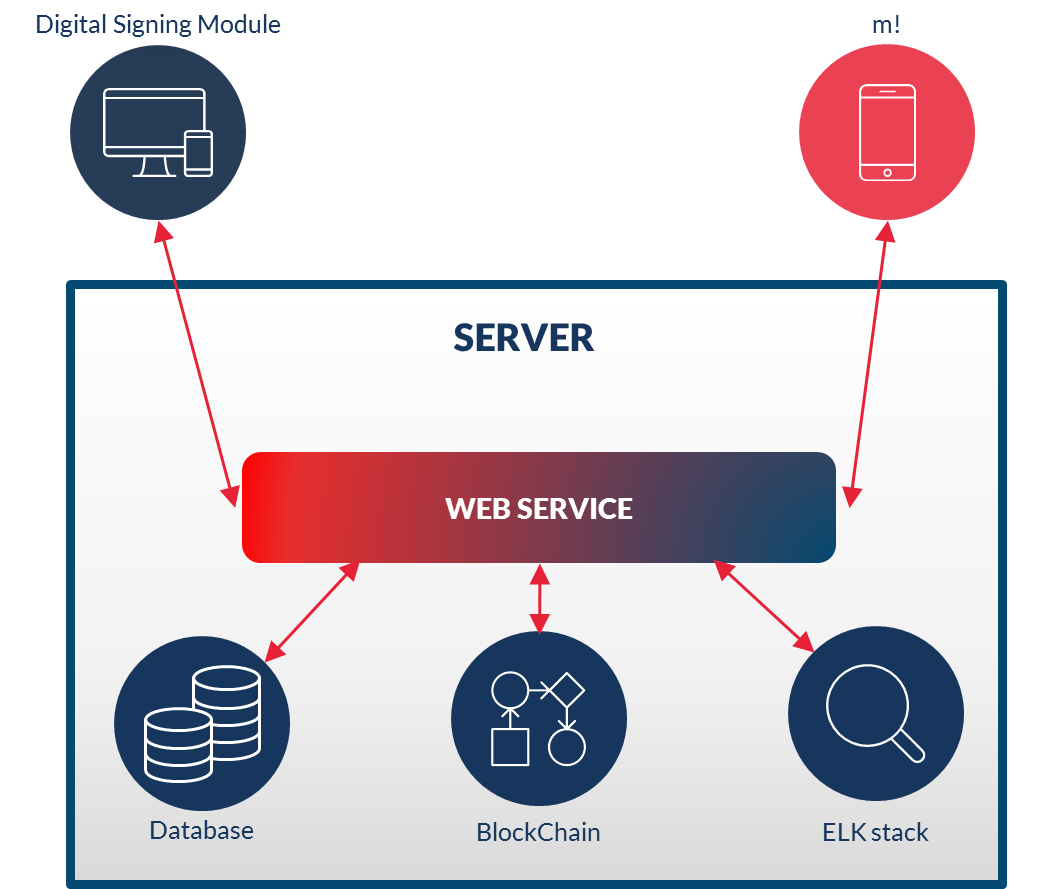
Web application with digital signing increases the speed at which documents can be reviewed and approved. Reducing time to product launch and downtime from correction of critical issues.
The ELK Stack provides users with a powerful platform that collects and processes data from multiple elements of MediqARC, stores that data in one centralized data store that can scale as data grows, and that provides a set of tools to analyse the data effectively.
Full text search means that you can find where document numbers, procedure titles, and other specific terms have been used within your entire technical documents or SOPs with a single search when there is a need for revision or investigation.
Private Block Chain the recording of transactions, maintained across several computers that are linked in a peer-to-peer network
MediqARC gives access to all information on a controlled basis ensuring documentation control for drafts, current and archived documents.
Mobile access via M! platform for secure communication allowing those on the move to sign documents permitting checking or entering information such as batch details or Non-conformance when away from your desk in the warehouse or factory.

Document organisation automatically archives documents when new versions are released and prevents accidental deletion of records required for regulatory compliance.
Notifications and reminders such as calibration requirements, CAPAs and certificate renewals are automatically triggered and sent to appropriate users.
Text Watermarking helps with control of documents which are automatically watermarked “Archive” when they cease to be current. When documents are printed they are automatically watermarked “uncontrolled” and prior to approval documents are watermarked “Draft”.
Documents are created within MediqARC or existing documents can be uploaded. Any existing document can instantly integrate into MediqARC system.
Documents can be printed and emailed directly from MediqARC.
Digital Signing allows efficient authorisation of documents to speed the quality and regulatory process.
Access to and control of data is provided by allocation of roles, responsibilities and controlled access via password.
Filtering and sorting of documents helps analyse for event reporting trend analysis etc.

QMS and CE certificates can be filed so that the current copy is immediately available and can trigger prompts when renewal is pending.
Quality manual and SOPs are always available at all required locations.
Audits and Management Reviews can be fled with controlled access to help with subsequent actions.
Training records can be filed with controlled access so managers have the immediate access to their reportees training records.
Suppliers Records are available online and instantly updated if a CAPA is allocated to the supplier. Prompts are triggered when a supplier assessment requires periodic review.
Calibration and maintenance records can trigger prompts when activities are required
Technical Files and Design Files can be filed within MediqARC providing the benefits of document control automatically operating version control as well as “Draft”, “Archive” and “Uncontrolled” watermarks
CAPAs, Customer Complaints and Non-conformities can all be controlled within MediqARC.
CAPAs are generated with MediqARC allowing actions to be automatically emailed to appropriate persons and signed digitally at each stage of implementation.
Quality Records such as final inspection reports can be filed within MediqARC providing the benefits of document control and searching.
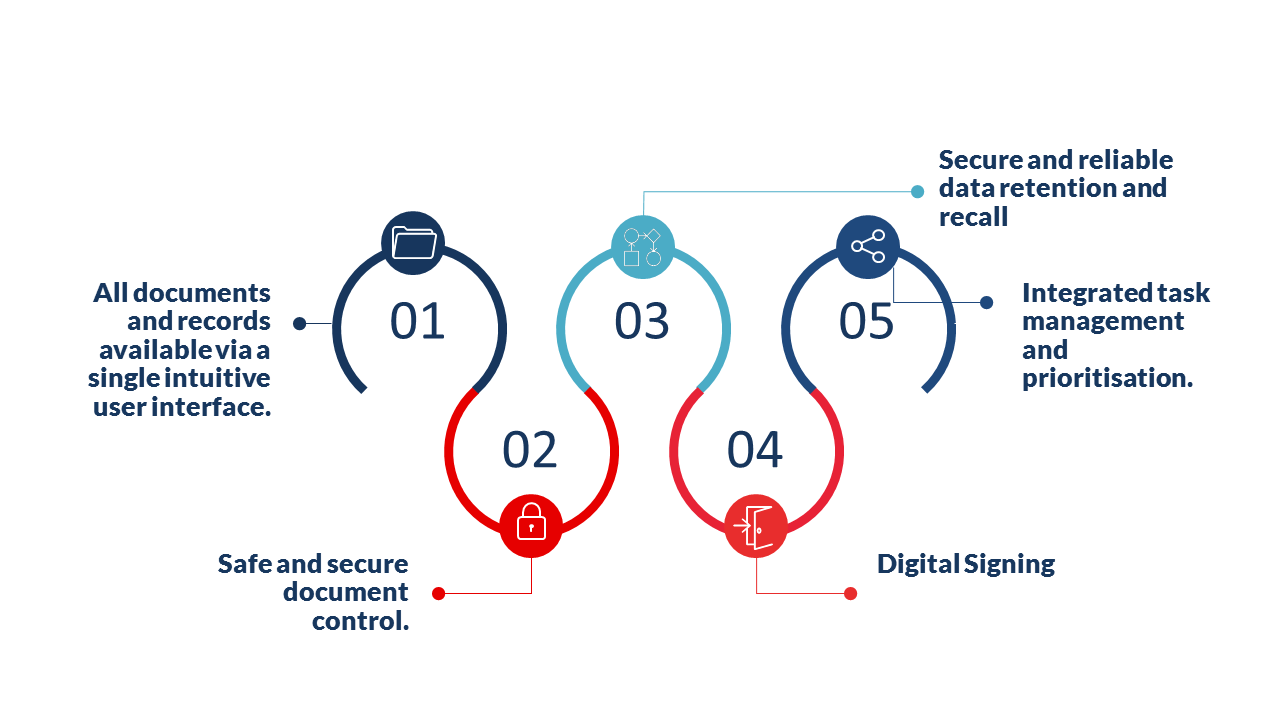
The global access from a common point of entry to all quality and regulatory compliance documents and records avoids ambiguity and lost documentation and allows multi-site companies to operate as a single entity.
Being able to control access through allocation of roles and responsibilities helps ensure that your documents while available to those who require access while remaining safe and secure.
Automatic and controlled archiving ensures that viewed documentation is current while allowing secure viewing and recall of archived documents to meet regulatory requirements for record retention.
Digital signing of documents by the required signatory’s irrespective of location, speeds you product introduction and eliminates the need to sign by hand and scan documents.
Tasks are flagged so that activities such as audits, calibration and validations are completed on time and non-conformities and recalls are avoided.

A single intuitive user interface for all your quality and regulatory needs. From Quality Manual to batch release records, including documents such as CAPA, Supplier Assessments, Training Records, and Audit Reports and many more to cover your entire quality and regulatory needs.
Expand from a start-up to a multinational organisation from a single device to an extensive product range. Can be tailored to meet individual unique requirements and terminology.
Can be expanded from the original regional requirement to cover your global needs.
Can be used by all levels of employees within the organisation with their access tailored to meet their needs and to prevent erroneous access, making data secure from deletion or unauthorised changes.

From a single common entry screen all information required for Quality and Regulatory compliance can be accessed through multiple routes while restricting access by user name and password.
Access Rights are controlled ensuring security of Intellectual Property and personal data
Document control within MediqARC ensures current Documents are available while allowing controlled access to archived documents for investigation and record retention.
Draft documents can be circulated for approval then transferred to current with previous version being archived. As documents change classification the appropriate watermark is added or removed.
Automatic archiving allowing controlled access to old documents for investigation and record retention for regulatory requirements.
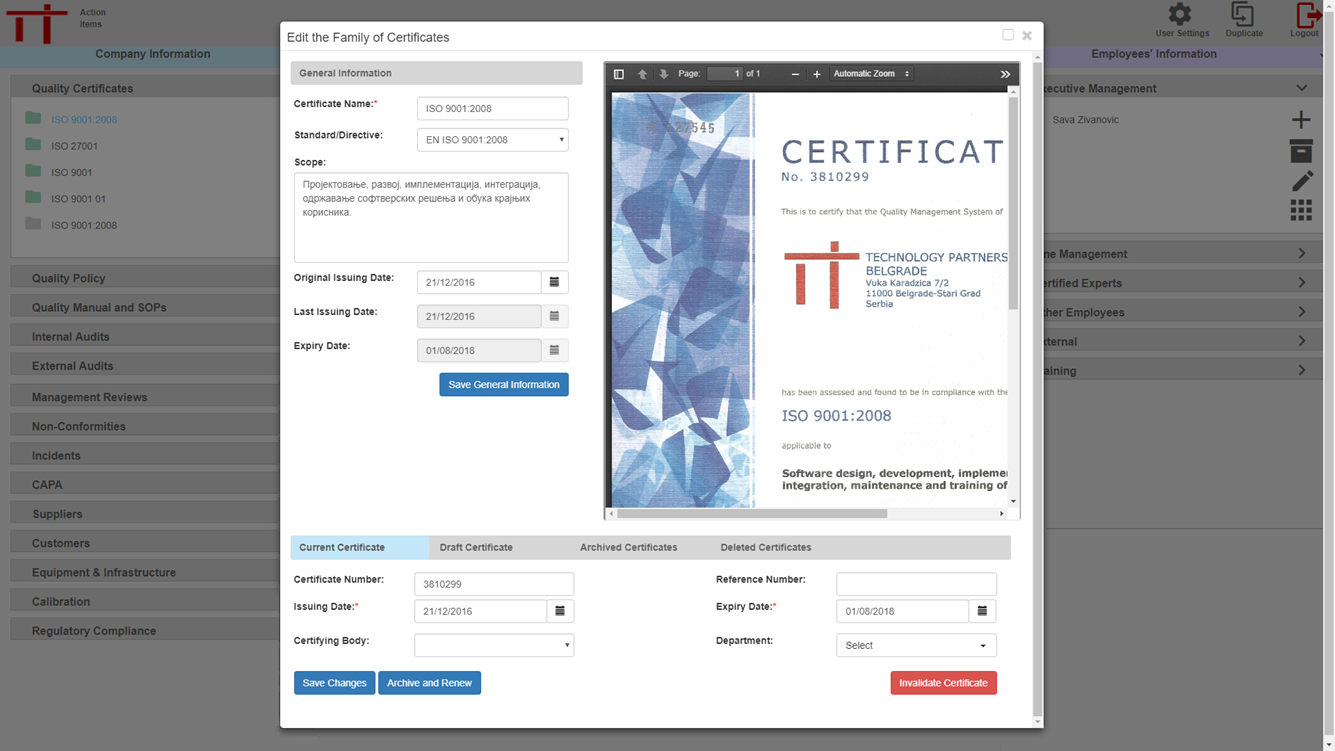
All certificates can be made available and controlled including draft, current and archived versions.
With draft, current and archived versions of certificates available a full change history is available and regulatory requirements for record retention is automatically achieved.
Current controlled copies of certificates can be emailed directly to customers.
Expiry dates are monitored automatically and notification of pending expiry is automatically indicated on the user interface of those responsible for the required actions.
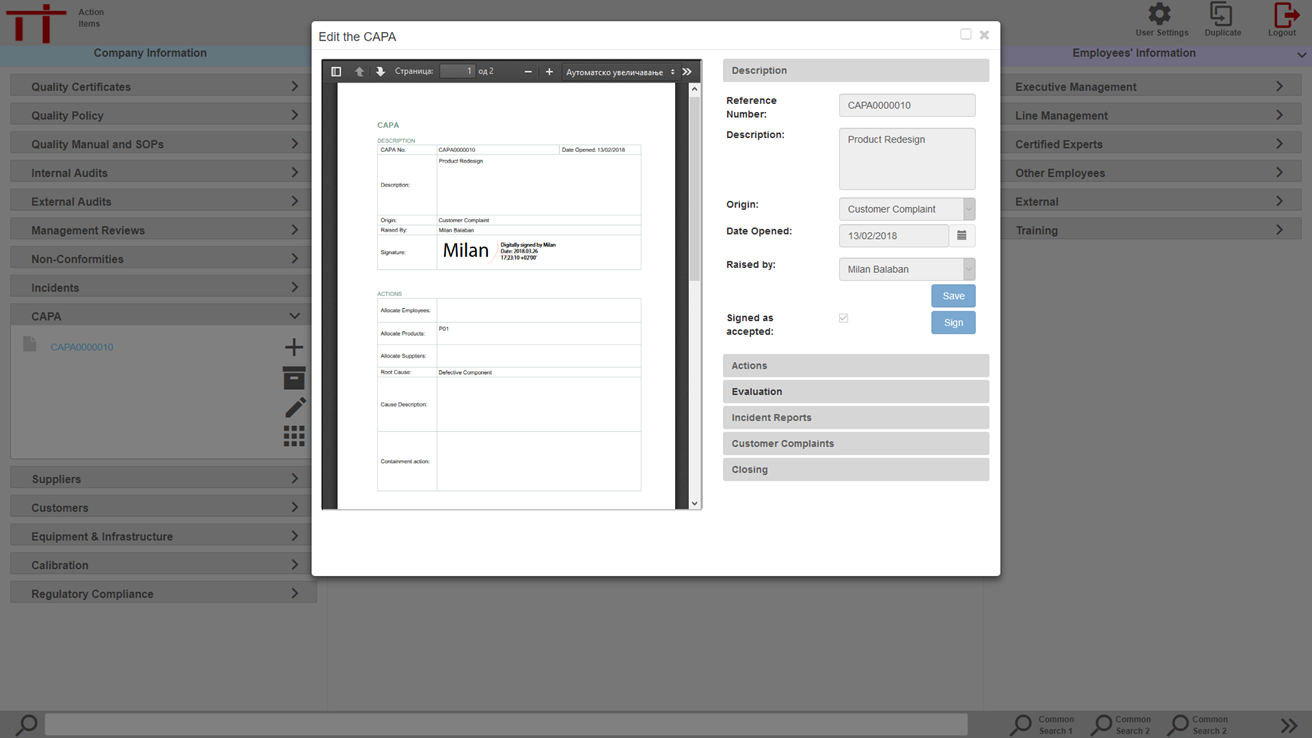
CAPAs can be generated within MediqARC and signed digitally.
At any stage the CAPA can be exported as a signed PDF document for circulation or emailing to external sources and suppliers. The digitally signed PDF meets the requirements for record retention.
Actions can be tracked and progressed through the MediqARC application with onscreen prompts when action is required.
At each stage the CAPA requires to be digitally signed before it can progress to the next stage of implementation or evaluation.
In electronic form the CAPA can be linked to a product, employee, department and suppliers. This facility is ideal for identifying trends and meeting the requirements of QMS/GMP systems.
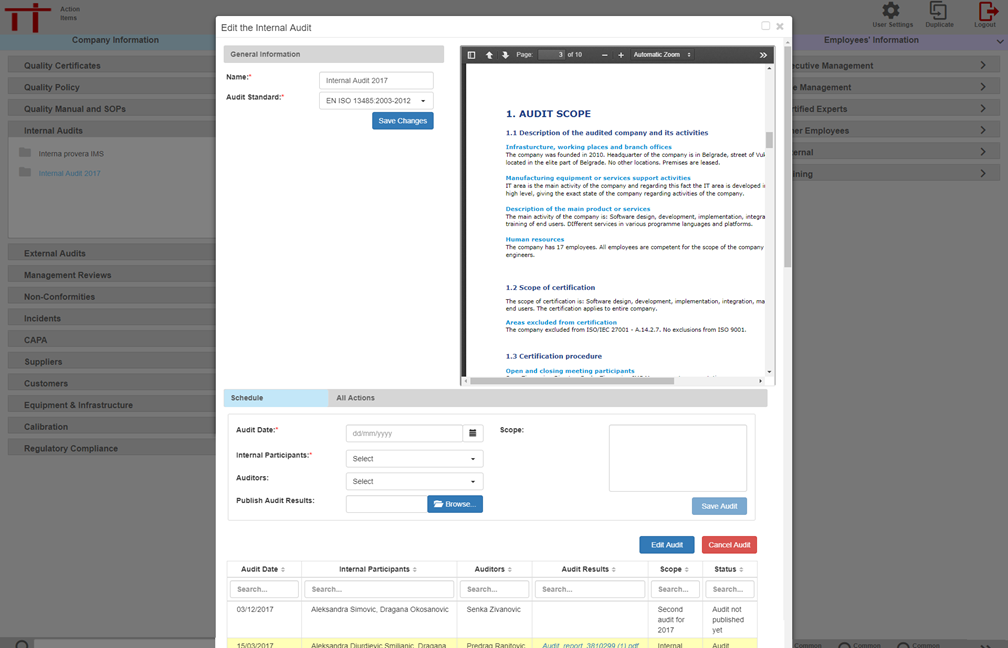
MediqARC will monitor and maintain the scheduling of internal audits and management reviews. Prompts will be sent to the appropriate person along with agendas or minutes.
Schedules can be updated, amended and added to at any stage.
CAPAs can be launched from audits or management reviews and monitored through the CAPA module.
Colour coded prompts of audits, meetings and CAPAs will appear on appropriate users interface screen.
Agendas, minutes and audit reports can be printed and emailed from the MediqARC application
Emailed notifications of meetings can be sent.
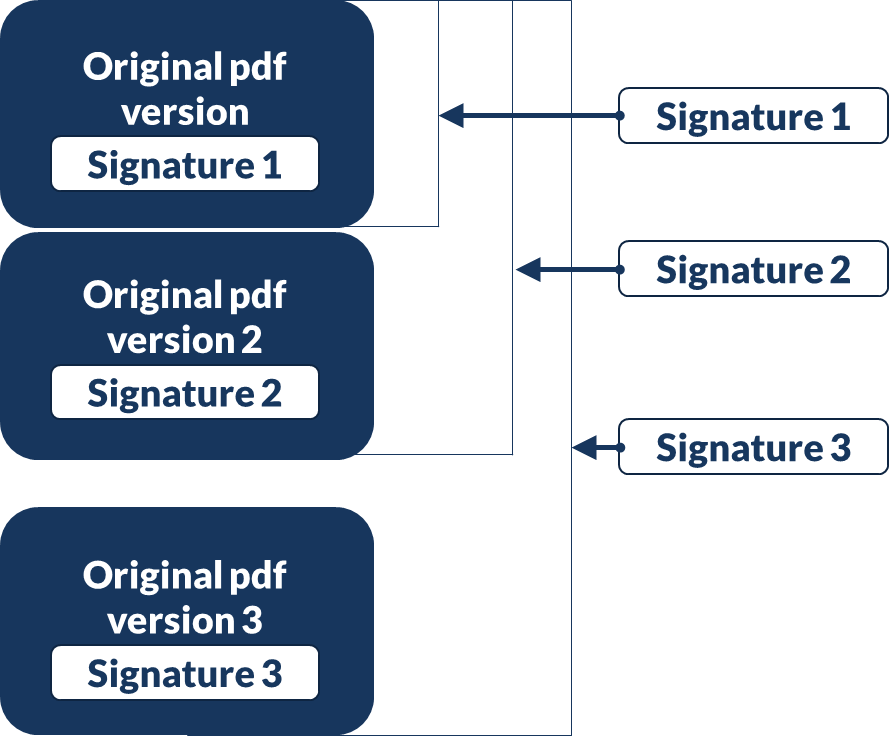
Documents can have single or multiple signatures as required.
Digital signatures are instantly verifiable on screen
Signatures are FDA 21 CFR Part 11 compliant making them suitable for use in the USA

31 Braintree Business Park
Blackwell Drive, Braintree
Essex, CM7 2PU, UK
T: +44 (0)1376 342 303